‘Absolutely remarkable’: No one who got Moderna’s vaccine in trial developed severe COVID-19 (sciencemag.org)

Science’s COVID-19 reporting is supported by the Pulitzer Center and the Heising-Simons Foundation.

Science’s COVID-19 reporting is supported by the Pulitzer Center and the Heising-Simons Foundation.

One of the experimental drugs that President Trump received while he was battling the coronavirus has been approved for emergency use by the Food and Drug Administration. The drug, made by the biotech company Regeneron, is the second antibody treatment to win emergency use approval from the FDA.
The treatment combines two antibodies — casirivimab and imdevimab — and administers them together by IV. In a clinical trial of about 800 people, the combination was shown to significantly reduce virus levels within days of treatment.

(CNN)The US Food and Drug Administration has issued an emergency use authorization for the first self-test for Covid-19 that can provide rapid results at home.

(Reuters) - Moderna Inc’s experimental vaccine is 94.5% effective in preventing COVID-19 based on interim data from a late-stage trial, the company said on Monday, becoming the second U.S. drugmaker to report results that far exceed expectations.
Together with Pfizer Inc’s vaccine, which is also more than 90% effective, and pending more safety data and regulatory review, the United States could have two vaccines authorized for emergency use in December with as many as 60 million doses of vaccine available this year.
LONDON, Nov 9 (Reuters) - Many COVID-19 survivors are likely to be at greater risk of developing mental illness, psychiatrists said on Monday, after a large study found 20% of those infected with the coronavirus are diagnosed with a psychiatric disorder within 90 days.
Anxiety, depression and insomnia were most common among recovered COVID-19 patients in the study who developed mental health problems, and the researchers also found significantly higher risks of dementia, a brain impairment condition.

(CNN)President-elect Joe Biden and Vice President-elect Kamala Harris' newly appointed Transition Covid-19 Advisory Board is led by established public health officials and staffed by a mix of doctors and current and former government officials, some with high-profile media visibility.

(CNN)Scientists Ugur Sahin and Ozlem Tureci have dedicated their lives to the field of oncology and infectious diseases, and spent years pioneering personalized immunotherapy treatments for cancer.

The reopening of restaurants, gyms and hotels carries the highest danger of spreading Covid-19, according to a study that used mobile phone data from 98 million people to model the risks of infection at different locations.

A nasal antiviral created by researchers at Columbia University Vagelos College of Physicians and Surgeons blocked transmission of SARS-CoV-2 in ferrets, suggesting the nasal spray also may prevent infection in people exposed to the new coronavirus.
The compound in the spray—a lipopeptide developed by Anne Moscona, MD, and Matteo Porotto, PhD, professors in the Department of Pediatrics and directors of the Center for Host-Pathogen Interaction—is designed to prevent the new coronavirus from entering host cells.
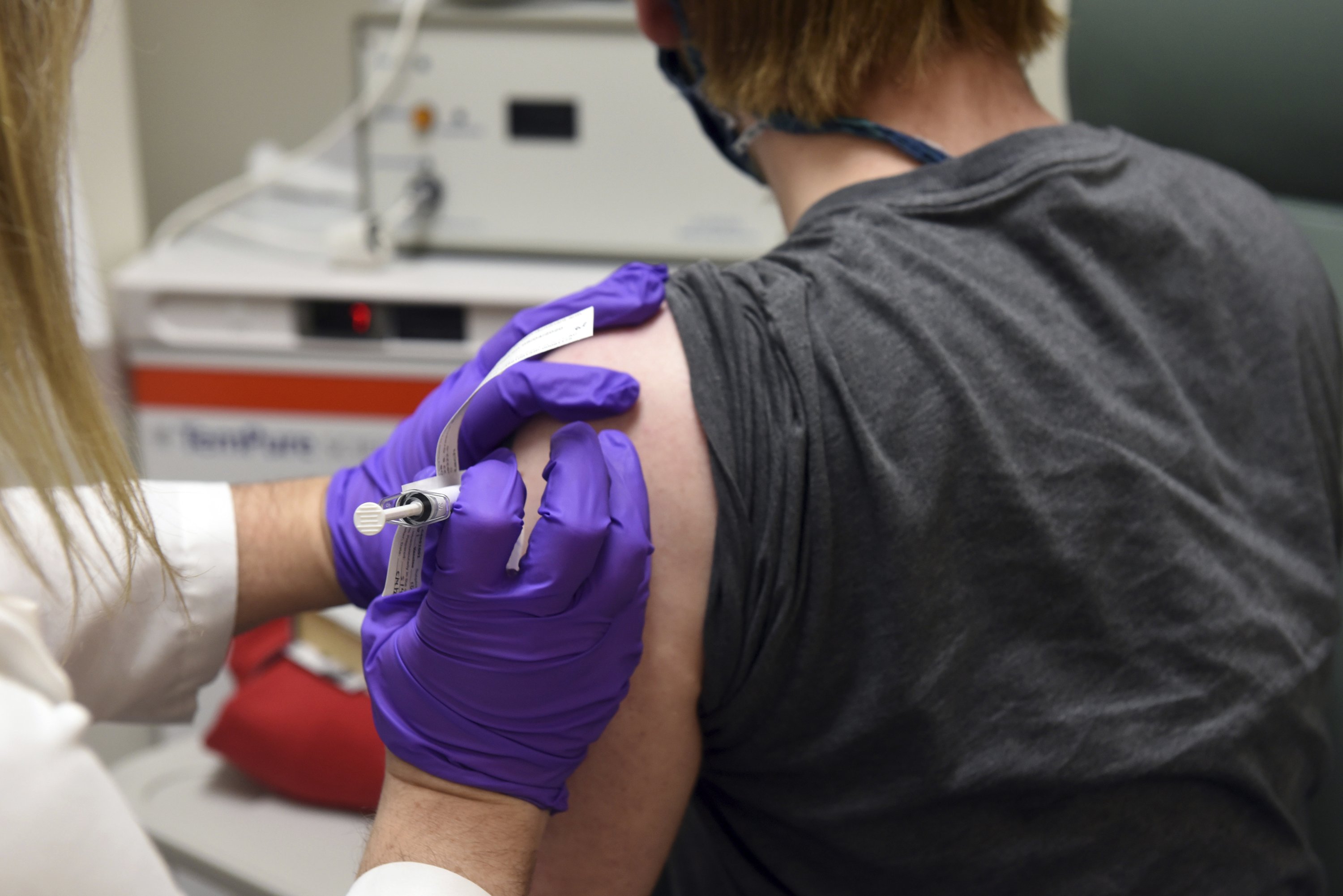
Pfizer, which is developing the vaccine with its German partner BioNTech, now is on track to apply later this month for emergency-use approval from the U.S. Food and Drug Administration, once it has the necessary safety information in hand.
Even if all goes well, authorities have stressed it is unlikely any vaccine will arrive much before the end of the year, and the limited initial supplies will be rationed.